ESLHO EQA Portal
Welcome! Below you can find information about the EQA schemes that ESLHO organizes with the support of the EuroClonality, EuroMRD, and EuroFlow consortia. Once logged in, you can register and manage your active EQA schemes.
EuroFlow test scheme

- Round 1
- Registration closed
- Round 2
- Registration closed
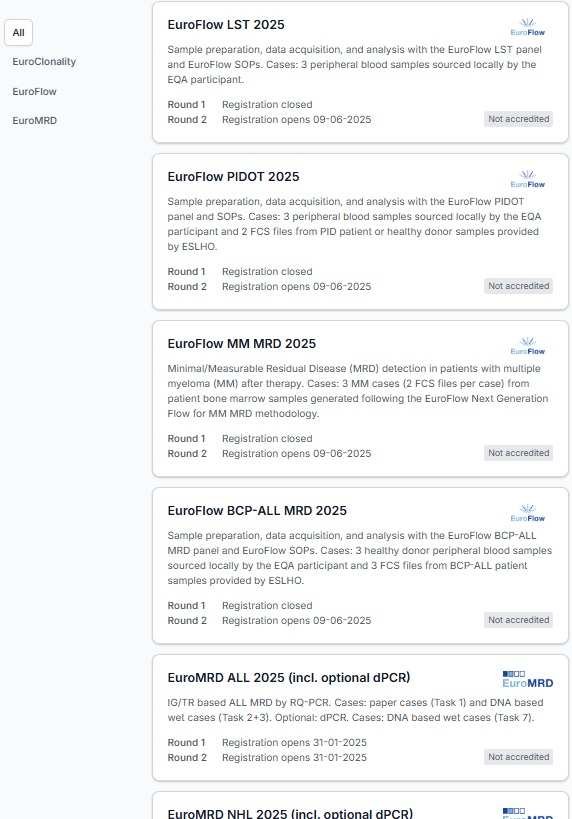
EuroFlow LST 2025

Sample preparation, data acquisition, and analysis with the EuroFlow LST panel and EuroFlow SOPs. Cases: 3 peripheral blood samples sourced locally by the EQA participant.
- Round 1
- Registration closed
- Round 2
- Registration closed
Not accredited
EuroFlow MM MRD 2025

Minimal/Measurable Residual Disease (MRD) detection in patients with multiple myeloma (MM) after therapy. Cases: 3 MM cases (2 FCS files per case) from patient bone marrow samples generated following the EuroFlow Next Generation Flow for MM MRD methodology.
- Round 1
- Registration closed
- Round 2
- Registration closed
Not accredited
EuroFlow PIDOT 2025

Sample preparation, data acquisition, and analysis with the EuroFlow PIDOT panel and SOPs. Cases: 3 peripheral blood samples sourced locally by the EQA participant and 2 FCS files from PID patient or healthy donor samples provided by ESLHO.
- Round 1
- Registration closed
- Round 2
- Registration closed
Not accredited
EuroFlow BCP-ALL MRD 2025

Sample preparation, data acquisition, and analysis with the EuroFlow BCP-ALL MRD panel and EuroFlow SOPs. Cases: 3 healthy donor peripheral blood samples sourced locally by the EQA participant and 3 FCS files from BCP-ALL patient samples provided by ESLHO.
- Round 1
- Registration closed
- Round 2
- Registration closed
Not accredited
ESLHO-EuroFlow PIDOT 2024

Sample preparation, data acquisition, and analysis with the EuroFlow PIDOT panel and SOPs. Cases: 3 local PB samples and 2 paper cases.
Not accredited