About ESLHO
The European Scientific foundation for Laboratory Hemato Oncology (ESLHO) is a scientifically independent foundation that promotes innovation, standardization, quality assessment, and education of laboratory diagnostics focused on lymphoid malignancies, together with the associated consortia EuroClonality, EuroFlow, and EuroMRD.
ESLHO EQA Program
ESLHO is taking over the main responsibility to organize the external quality assessment (EQA) schemes of EuroClonality, EuroFlow, and EuroMRD. As EQA provider of these schemes, ESLHO will continue to work with the expert labs that have initiated and organized these schemes in the previous two decades.
To manage all EQA schemes, ESLHO developed this website, the ESLHO EQA Portal.
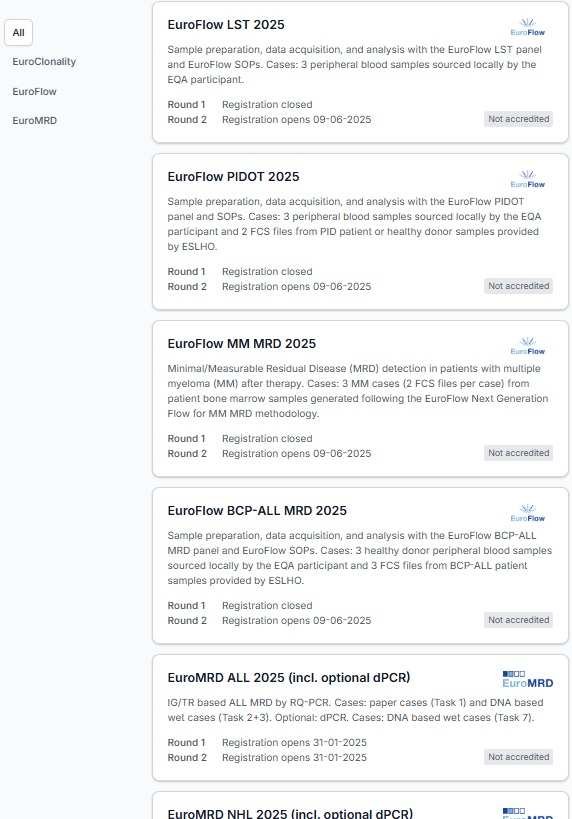
Detailed information about all schemes and upcoming rounds can be found on the ‘All Schemes’ page.


EuroClonality
EuroClonality is a European scientifically independent foundation aiming to promote continuous innovation and standardization of laboratory diagnostics in hemato-oncology with a special emphasis on clonality testing and supporting quality control and education in laboratory diagnostics. EuroClonality developed and standardized novel multiplex PCR assays for detection of immunoglobulin (IG) and T-cell receptor (TR) gene rearrangements. These assays are now widely used for the diagnosis of lymphoproliferative disorders.
Together with ESLHO, EuroClonality organizes the EuroClonality EQA scheme for IG/TR Clonality Testing in Suspected Lymphoproliferations/Lymphoma once per year.


EuroFlow
EuroFlow develops and standardizes fast, accurate, and highly sensitive flow cytometric tests for diagnosis and prognostic (sub)classification of hematological malignancies as well as for evaluation of treatment effectiveness during follow-up.
Together with ESLHO, EuroFlow organizes the EuroFlow LST, PIDOT, MM MRD, and BCP-ALL MRD EQA schemes twice per year.


EuroMRD
EuroMRD develops guidelines for the interpretation of RQ-PCR MRD and NGS MRD data. These guidelines ensure identical interpretation of MRD data between different laboratories that use the same MRD-based clinical protocol.
Together with ESLHO, EuroMRD organizes EQA schemes for quantitative MRD diagnostics for lymphoid malignancies twice per year for the EuroMRD sections ALL (incl. KMT2A), NHL, and Ph+ALL. Pilot schemes include dPCR and NGS based schemes.
ESLHO EQA Team

Final responsibility for the ESLHO EQA Program

EQA Officer EuroClonality and EuroMRD schemes (lead)
EQA Officer EuroFlow schemes
Internal auditor

EQA Officer EuroFlow schemes (lead)
Internal auditor
Technician

Technician
EQA Officer EuroClonality and EuroMRD schemes
Internal auditor

Quality Coordinator
EQA Officer EuroMRD schemes
Technician
Internal auditor

Internal auditor
Colophon
ESLHO EQA Portal
Version: 1.11.0
Last updated: April 16, 2026
General Terms and Conditions
Copyright © 2026 ESLHO. All rights reserved.
-
Design
Evers + de Gier
Rotterdam, The Netherlands -
Development
Jannes & Mannes
Rotterdam, The Netherlands